Scroll to:
Efficacy of differentiated targeted interferon and immunomodulatory therapy focused on pathological immunophenotypes in patients with atypical chronic active herpesvirus infections
https://doi.org/10.21886/2219-8075-2024-15-1-165-175
Abstract
Objective: In a prospective cohort study to evaluate the clinical and immunological efficacy of differentiated targeted interferon and immunomodulatory therapies focused on identified pathological immunophenotropes and associated clinical manifestations in immunocompromised patients suffering from atypical chronic active herpes viral infections (ACA-HVI).
Materials and methods: 335 patients suffering from mixed-AHA-HVI were examined. The study complex included: methods for detecting herpesviruses: serodiagnosis, PCR-RV; immunological methods: a research of subpopulation of blood lymphocytes (method of a flow cytometry), determination of the spontaneous and induced products of IFNα and IFNγ, levels of serum cytokines and immunoglobulins (IL-1β, IL-6, IL-17, TNFα and IFNα and IFNγ, Ig A, M, G) by ELISA. The study was approved by the ethics board and informed consent was obtained from all patients.
Results: Integral formulas of disorders in the antiviral immune defense system were created, which made it possible to isolate 3 pathological immunophenotypes (PIF): PIF1: NG↓+ind.IFNα/IFNγ↓+CTL↓+EKK↓+IgM↑+hypecytokinemiya (IL-1β↑+IL-6↑+TNFα↑); PIF2: NG↓+ ind. IFNα/IFNγ↓ + EKK↓+ IgG↓+hypercytokinemiya (IL-1β↑+IL-6↑+TNFα↑) and PIF3: NG↓+ind.IFNα/IFNγ↓+hypercytokinemiya (IL-1β↑+IL- 6↑+TNFα↑). Taking into account the identified disorders, a program of targeted interferon and immunomodulatory therapy was developed for each PIF: local and systemic IFN therapy + hexapeptide was developed for PIF1; for PIF2- local and systemic IFN therapy + glucosaminimuramyldipeptide; for PIF3-local and systemic IFN therapy.
Conclusions: High clinical efficacy was demonstrated in 100% of patients with three groups of ACA-HVI. Immunological effectiveness of targeted interferon and immunomodulatory therapy programs: 89.5% for PIF1; for PIF2-57.6% and for PIF3- 37.5% of cases.
For citations:
Nesterova I.V., Khalturina E.O. Efficacy of differentiated targeted interferon and immunomodulatory therapy focused on pathological immunophenotypes in patients with atypical chronic active herpesvirus infections. Medical Herald of the South of Russia. 2024;15(1):165-175. (In Russ.) https://doi.org/10.21886/2219-8075-2024-15-1-165-175
Introduction
A pressing challenge to this date is the growth of infectious diseases with atypical courses [1–3]. According to WHO data, herpesviral infections are attributed to uncontrolled infections and have assumed the character of pandemics, showing steady annual growth [3–7]. Currently, poorly studied atypical clinical forms of these infections, including atypical chronic active herpes infections (ACA-HVI), are increasingly being identified [8]. Translational immunology is based on the involvement of advances in immunological science for solving practical human problems to ensure high-efficiency health care delivery. In particular, one of the objectives of research conducted within the framework of translational immunology is related to the acquirement of new knowledge on the immunopathogenesis of ACA-HVI in order to form the basis for the development of new diagnostic approaches and the creation of differentiated methods for correcting impairments of antiviral immune defense [10]. Thus, obtaining new scientific data on the immunopathogenesis of ACA-HVI provides the basis for a practical solution to the current problem of improving the efficacy of treatment for patients with ACA-HVI.
The purpose of the study is to assess the clinical and immunological efficacy of differentiated targeted interferon and immunomodulation therapy oriented towards identified pathological immunophenotypes and associated with them clinical manifestations in immunocompromised patients suffering from ACA-HVI within the framework of a prospective cohort study.
Materials and methods
The study cohort included 335 patients suffering from mixed ACA-HVI, with a history of the disease ranging from 3 to 12 years mentioned here as the group of investigation (GI). The ACA-HVI was diagnosed in virtue of the presence of laboratory examinations and clinical signs of immunocompromise using the clinical criteria of the first and second order previously developed by the authors of this study [11].
A comprehensive examination included traditional methods, in particular collecting complaints and anamnesis, physical examination, general clinical laboratory tests, etc. Methods for serological diagnosis of HVI included the determination of IgM and IgG antibodies to different Epstein Barr Virus antigens (VCA, EBNA, NA), cytomegalovirus (CMV), herpes simplex virus HSV-1/2 type (HSV), and human herpes virus type 6 (HHV) by ELISA using test systems from NPO Diagnostic Systems (Russia), and by molecular genetic research methods using RT-PCR (the test system “AmpliSens” (Russia) for detecting the genome of herpes viruses in biomaterials (blood, saliva, urine, scrapings from the tonsils and posterior pharyngeal wall).
The scope of the immunological study included the determination of spontaneous and induced production of IFNα and IFNγ, levels of serum cytokines and immunoglobulins including IL-1β, IL-1RA, IL-6, IL-17A, TNFα and IFNα and IFNγ, Ig A, Ig M, and Ig G by ELISA (test systems of ZAO Vector-Best, Novosibirsk; LLC Cytokin, St. Petersburg), and the determination of the subpopulation composition of blood lymphocytes by the flow cytofluorometry method (Beckman Coulter International, USA).
The comparison group consisted of 30 conditionally healthy individuals commensurable in gender and age to GI patients.
The study was approved by the ethics committee; all patients gave informed consent to participate in the study and to process personal data, in accordance with the World Medical Association Declaration of Helsinki (WMA Declaration of Helsinki, 2013).
For the statistical processing of the obtained data, the Microsoft Excel computer software was used. The results were presented as median and upper and lower quartiles (Me [Q1;Q3]). Comparative statistics included calculations of the Mann-Whitney and Wilcoxon tests. Differences were considered significant at p<0.05.
Results
Upon the investigation of the clinical progression features of herpesvirus infections in patients of the GI, the previously developed clinical criteria for signs of immunocompromise of the first and second order [11] were used. In accordance with the first-order clinical criteria, all patients of the GI had a high relapse frequency of HVIs, which emerged more than 6 times per year, and a high frequency of recurrent acute respiratory viral infections (ARVIs), specifically patients had more than 7 episodes per year, often from 10 to 20 episodes per year or 1–2 episodes per month. In these patients, relapses of HVIs were clinically predominantly uncomplicated but long-lasting and difficult to treat with standard therapy. The duration of the period of clinical manifestations of ARVI ranged from 5 to 10 days, more specifically 7.5 (7.0; 8.0) days, while the duration of the clinically safe period did not exceed 6.3 months per year. In addition, recurrent ARVIs were often complicated by the accession of a bacterial infection causing acute diseases of the upper and lower respiratory tract including acute bronchitis, acute pneumonia, etc., and ENT organs including acute sinusitis, acute purulent adenoiditis, etc.
It should be noted that among the pathognomonic clinical manifestations, a prolonged feeling of severe asthenia and symptoms of chronic fatigue syndrome (CFS) were most characteristic for patients with ACA-HVI; patients were bothered by sweating, intermittent sore throat, migrating pain and discomfort in the muscles, and diarthroses known as fibromyalgia and arthralgia, as well as by cephalgia, prolonged subfebrile fever, regional lymphadenopathy, disturbance of the sleep structure and the process of falling asleep, decreased memory, attention, intelligence, and less often psychogenic depression. In some patients suffering from ACA-HVI, clinical manifestations of peripheral neuro- and sensoropathies, myalgia and arthralgia with signs of fibromyalgia, pronounced and progressive cognitive and mnestic disorders were also revealed. All these signs taken together indirectly indicated the development of neuroimmunoinflammation with localization of the process in both peripheral and central parts of the nervous system. These symptoms, related to the clinical criteria signs of immunocompromise of the second order, were assessed using a 5-point scale developed by the authors to assess the severity of the criterion signs/symptoms of CFS and cognitive disorders [11]. The severity of symptoms on this scale in mixed ACA-HVI was 54.5 [ 46.75; 62.25] points.
The assessment of the indicators, which specified the immune system state in patients with ACA-HVI, identified different defects in the functioning of the antiviral immune defense, which made it possible to clarify a number of features of this infection immunopathogenesis. They included the deficiency of IFNα and IFNγ, which manifested itself as the impairment of induced production and a decrease in the concentration in the blood serum, as well as deficiency of natural killer cells (NKs or NKCs), cytotoxic T lymphocytes (CTLs), disimmunoglobulinemia, and hypercytokinemia of proinflammatory cytokines. A detailed examination and further specification of the variability of different combinations of these impairments made it possible to identify three main pathological immunophenotypes.
The first immunophenotype was specified by the following defects in the immune system: the impairment of the induced production of IFNα and IFNγ; deficiency of CTLs (CD3+CD8+) in 76.4% of cases; NK deficiency in 36.6% of cases; neutropenia, more specifically deficiency of neutrophil granulocytes (NGs) in 83.7% of patients, as well as hyperproduction of all studied pro-inflammatory cytokines and increased levels of IgM class antibodies in 84.2% of patients. Significant defects in the interferon system were manifested by the impairment of the induced production of IFNα in 100% of cases and IFNγ in 48% of patients, along with a decrease in the level of spontaneous production of IFNα by 1.7 times and IFNγ by 1.6 times in relation to the group of conditionally healthy individuals. Thus, the integral formula for disorders in the antiviral immune defense system for this immunophenotype is as follows: NG↓ + induced IFNα/IFNγ↓ + CTL↓ + NK↓ + IgM↑ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑). The clinical features of patients assessed by the criteria for immunocompromise of the 1st and 2nd order were the following: the frequency of HVI relapses was 10.0 (9.5;12.5) cases per year; the duration of HVI relapses amounted to 8.0 (6.5;10.5) days; the duration of the period of clinical well-being (inter-relapse period) was 6.5 (4.5; 8.5) days; the frequency of ARVI was 11.0 (8.5; 13.5) episodes per year; the duration of ARVI was 7.5 (6.5; 19.5) days. Assessing clinical criteria signs for immunocompromise of the second order according to the “Scale for assessing clinical criteria/signs of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) and cognitive disorders” showed that the number of points scored amounted to 54.5 [ 46.75; 62.25] with a maximum score of 65.0 points.
The second immunophenotype was specified by secondary interferonopathies, which included the deficiency of the induced production of IFNα and IFNγ, the deficiency in the number of NGs (secondary neutropenia), and the deficiency of NKs. The investigation of the functioning of the interferon system in these patients identified significant defects, manifested by the impairment of the induced production of IFNα in 100% of cases and IFNγ in 38% of patients, along with a decrease in the level of spontaneous production of IFNα in 21.4% and IFNγ in 8.4% of patients of this group. A significant decrease in the absolute content of CD3‒CD16+CD56+ lymphocytes was shown in 76.6% of cases, and CD3+CD8+ subpopulation in 41.1% of cases, which indicated the incompetence of antiviral immune defense against pathogens with clinical manifestations in the form of frequent relapses of HVI and the recurrent ARVI incidence. Against the background of no changes in the absolute values of CD3‒CD19+ lymphocytes, normal levels of IgA and IgM (p<0.05) and a decrease in the concentration of IgG by 1.2 times (p<0.05) were revealed in the blood serum; that may attest to a deficiency of antibody immune response to pathogens. This group of patients was also characterized by the overproduction of all proinflammatory cytokines studied. The integral formula for impairments in the antiviral immune defense system for the second immunophenotype was as follows: NG↓ + induced IFNα/IFNγ↓ + NK↓ + IgG↓ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑). The clinical features of patients assessed using the criteria for immunocompromise of the 1st and 2nd order were represented by the following items: the frequency of relapses of HVI was 10.0 (8.0;14.5) cases per year; the duration of relapses of HVI was 8.0 (6.5;10.5) days; the duration of the period of clinical well-being (inter-relapse period) amounted to 6.5 (4.5; 8.5) days; the frequency of ARVI was 11.5 (10.5; 14.5) cases per year, and the duration of ARVI amounted to 7.5 (6.5; 10.5) days. Upon assessing clinical criterion signs of the immunocompromise of the second-order according to the “Scale for assessing clinical criteria/signs of CFS/ME and cognitive disorders,” the number of points corresponded to 46.0 [ 33.0;55.5] with a maximum score of 65.0 points.
Patients with the third immunophenotype were specified by isolated impairments in the interferon system, manifested by a decrease in the concentration of serum interferon of both types and dropping in the induced production of predominantly IFNα. The identified impairments in the interferon system were in the nature of significant defects and were manifested by disturbances of the induced production of IFNα in 100.0% of cases and IFNγ in 85.7% of patients, as well as by a decrease in the level of spontaneous production of IFNα in 100.0% and IFNγ in 79.5% of patients of this group. It should be noted that the quantitative determination of NGs revealed that the prevalence of neutropenia in the patients of this GI was about 18% and was significantly lower compared to the immunophenotypes described above. No significant differences in the indices of cellular and humoral immunity in comparison to the control group were revealed. However, the overproduction of all studied indices was found during the investigation of pro-inflammatory cytokines. The integral formula for disorders in the antiviral immune defense system of the third immunophenotype was as follows: NG↓ + induced IFNα/IFNγ↓ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑). Assessing the clinical features of patients with this immunophenotype in accordance with the clinical criteria for immunocompromise of the 1st and 2nd order revealed the following peculiarities: frequency of HVI relapses was 12.0 (10.5; 14.5) cases per year; the duration of HVI relapses was 9.5 (7.5; 12.5) days; the duration of the period of clinical well-being (inter-relapse period) reached 3.0 (2.5; 5.5) days; the frequency of ARVI was 12.5 (10.5; 14.5) cases per year; the duration of ARVI reached 8.5 (5.5; 10.5) days. When assessing clinical criteria signs for immunocompromise of the second order according to the “Scale for assessing clinical criteria/signs of CFS/ME and cognitive disorders,” the number of points corresponded to 52.5 [ 45.0; 50.5] with a maximum score of 65.0 points.
Basing on the studied and described clinical characteristics of patients with ACA-HVI, as well as considering the identification of one of the three dominant pathological immunophenotypes resulting from clinical and immunological examination, which were determined by the nature and severity of impairments in the immune and interferon systems, three programs of differentiated targeted interferon and immunomodulation therapy were developed (Table 1).
Таблица / Table 1
Программы дифференцированной таргетной
интерфероно- и иммуномодулирующей терапии
Targeted interferon and immunomodulatory therapy programs
|
Молекулярные и клеточные механизмы терапии Molecular and cellular mechanisms of therapy |
Программа 1 Program 1 |
Программа 2 Program 2 |
Программа 3 Program 3 |
|
Системная интерферонотерапия Для восстановления уровня индуцированной продукции ИФН альфа и ИФН гамма, количества ЕКК рекИФНα2b — в комбинации с антиоксидантами препарат «Виферон®» в форме суппозиториев по схеме: 3 млн МЕ в день в течение 1 месяца, далее 2 млн МЕ в день (1 мес.), далее 1 млн МЕ (1мес.), далее 1 млн МЕ в режиме 3 раза в неделю в течение 2 недель, далее в дозе 1 млн МЕ дважды в неделю в течение 2 недель. Общий курс — 4 месяца. Systemic interferon therapy: to restore the level of induced production of IFN alpha and IFN gamma, the amount of EКК: reсIFNα2b in combination with antioxidants — the preparation "Viferon®" in the form of suppositories according to the scheme: 3 million IU per day for — 1 month, then 2 million IU per day — 1 month, then 1 million ME — 1 month, then 1 million IU in the mode 3 times a week for 2 weeks; then at a dose of 1 million IU twice a week for 2 weeks. The total course was 4 months. |
|
|
|
|
Локальная интерферонотерапия «Виферон®»-гель в виде аппликаций на слизистую оболочку носовых ходов, заднюю стенку глотки и дужки миндалин. Local interferon therapy "Viferon®" gel in the form of applications on the mucous membrane of the nasal passages, the back wall of the pharynx and the arms of the tonsils. |
|
|
|
|
Иммуномодулирующая терапия Для восстановления ЦТЛ и EKТ — препарат «Имунофан®» 45 мкг/мл 1 раз в день, в/м. Три десятидневных курса с перерывом в 20 дней в течении 3 месяцев. Курсы препаратов Имунофан и Изопринозин чередовали с перерывом 5 дней между курсами. Immunomodulatory therapy For the recovery of CTL and EKT — Imunofan® 45 μg/mL 1 once a day, IM. Three ten-day courses with a break of 20 days for 3 months. The courses of Imunofan and Isoprinosine were alternated with a break of 5 days between courses. |
|
|
|
|
Для восстановления НГ и ЕКК — глюкозаминилмурамилдипептида (ГМДП) «Ликопид» 3мг/сут. 3 10-дневных курса ежемесячно в течении 3 месяцев в перерывом в 20 дней. Курсы препаратов Ликопид® и Изопринозин чередовали с перерывом 5 дней между курсами. For the recovery of NG and EKK — glucosaminylmuramyl dipeptide (GMDP) "Lycopide" 3 mg/day three 10-day courses monthly for 3 months at a break of 20 days. Courses of Lycopid® and Isopinosine were alternated with a break of 5 days between courses. |
|
|
|
|
Противовирусная терапия Фамцикловир (препарат Фамвир®) 1500 мг/сут., длительность приёма 10 дней 1 раз в месяц или Ацикловир/Валцикловир в дозе 1500 мг в день в течение 10 дней при инфекции, вызванной ВПГ 1и 2 типов. А также Инозин пранобекс («Изопринозин») 1500 мг/сут., ежедневно в течение 10–14 дней с повторением 2–3 курсов с интервалом в 20 дней. Общая длительность терапии — 3 мес. Antiviral therapy Famciclovir (Famvir®) 1500 mg/day is the duration of administration of 10 days 1 times a month or Aciclovir/Valciclovir at a dose of 1500 mg per day, for 10 days in case of infection caused by HSV of types 1 and 2. As well as Inosine pranobex ("Isoprinosine") 1500 mg/day, daily for 10-14 days with a repetition of 2–3 courses with an interval of 20 days. The total duration of therapy is 3 months. |
|
|
|
The efficacy of each developed program was assessed using the previously proposed diagnostically significant immunological criteria including the determination of serum levels of IL-1β, IL-17A, TNFα, serum IFNα, IFNγ, and the proportion of NKCs (CD16+CD56+) among peripheral blood lymphocytes of patients with AHA-HVI.
In assessing the immunological efficacy of differentiated targeted interferon and immunomodulation therapy programs, the authors relied on the technique of determining the trend of changes in indicators relative to their initial levels before the start of therapy. A positive trend in changes in each of the studied blood parameters of a patient with ACA-HVI during treatment was assessed as 1 point. The following was taken as a positive trend:
- for pro-inflammatory cytokines, the positive trend was a decrease in their concentration in peripheral blood serum by at least 1.5 times;
- for interferons α and γ, the positive trend was an increase in their concentration in peripheral blood serum by at least 1.5 times;
- for the rate of NKs, the positive trend was an increase in the number of NKs in the peripheral blood by at least 1.5 times.
Based on these prerequisites, the following values were analyzed:
- the values of each indicator before and after treatment in absolute values and results of statistical assessment of these shifts using the Mann-Whitney test (Table 2);
- the proportion of positive changes (% efficacy) in each indicator during the treatment process and statistical comparison of treatment results on different programs with the χ² criterion (Table 2);
- the rates of deviations of the shift value of each indicator before and after treatment from the control (Figs. 1 and 2); the control values for each indicator are presented in Table 3;
- a total assessment in points for shifts of all 6 indicators within each treatment program and comparison of their 95% confidence intervals for each program with the constructed ROC curves of their diagnostic significance (Fig. 3).
Таблица / Table 2
Сравнительная иммунологическая эффективность программ
таргетной интерфероно- и иммуномодулирующей терапии
Comparative immunological efficacy
of targeted interferon and immunomodulatory therapy programs
|
Критериальный показатель Criteria indicator |
Медиана [минимум; максимум] Median [minimum; maximum] |
р1 р2 р3 |
|||
|
Программа 1 Program 1 (n = 156) |
Программа 2 Program 2 (n =100) |
Программа 3 Program 3 (n = 79) |
|||
|
ИЛ-1β, пг/мл IL-1β, pg/mL |
До лечения Before therapy |
69,0 [ 34,2; 100,8] |
86,9 [ 68,3; 127,3] |
96,2 [ 65,2; 165,8] |
- - - |
|
после лечения after therapy |
39,5 [ 21,7; 73,3] |
59,7 [ 34,7; 87,4] |
58,6 [ 14,7; 87,4] |
<0,001* <0,001* <0,001* |
|
|
% эффективности % efficiency |
79,2% |
25% |
87,5% |
<0,001* 0,009* 0,226 |
|
|
ИЛ-17A, пг/мл IL-17A, pg/mL |
До лечения Before therapy |
0,60 [ 0,44; 0,69] |
0,58 [ 0,45; 0,69] |
0,64 [ 0,20; 0,91] |
- - - |
|
После лечения After therapy |
0,56 [ 0,39; 0,71] |
0,63 [ 0,40; 0,71] |
0,61 [ 0,21; 0,75] |
0,204 0,052 0,223 |
|
|
% эффективности % efficiency |
100% |
100% |
100% |
1,000 1,000 1,000 |
|
|
ФНОα, пг/мл TNFα, pg/mL |
До лечения Before therapy |
29,3 [ 16,1; 48,7] |
27,6 [ 16,1; 48,7] |
28,1 [ 13,3; 48,7] |
- - - |
|
После лечения After therapy |
29,9 [ 13,9; 39,5] |
34,0 [ 19,5; 48,7] |
34,0 [ 20,3; 48,7] |
0,726 0,065 0,149 |
|
|
% эффективности % efficiency |
41,7% |
20,8% |
25% |
0,123 0,226 0,734 |
|
|
ИФНα, пг/мл IFNα, pg/mL |
До лечения Before therapy |
17,8 [ 10,2; 26,7] |
20,2 [ 14,1; 25,4] |
13,4 [ 2,1; 48,6] |
- - - |
|
После лечения After therapy |
34,1 [ 23,9; 56,2] |
26,8 [ 12,5; 45,4] |
18,6 [ 7,1; 45,2] |
<0,001* 0,004* 0,190 |
|
|
% эффективности % efficiency |
100% |
54,2% |
37,5% |
<0,001* <0,001* 0,252 |
|
|
ИФНγ, пг/мл IFNγ, pg/mL |
До лечения Before therapy |
29,8 [ 15,8; 46,1] |
25,0 [ 18,4; 32,4] |
36,6 [ 10,8; 45,9] |
- - - |
|
После лечения After therapy |
62,5 [ 33,2; 101,2] |
43,2 [ 39,4; 49,7] |
43,3 [ 23,5; 49,7] |
<0,001* <0,001* <0,001* |
|
|
% эффективности % efficiency |
87,5% |
100% |
91,7% |
0,077 0,640 0,153 |
|
|
Естественные киллеры, % EKK % |
До лечения Before therapy |
8,0 [ 5,2; 21,2] |
5,1 [ 2,0; 11,0] |
12,2 [ 9,2; 20,0] |
- - - |
|
После лечения After therapy |
10,2 [ 4,8; 16,5] |
6,8 [ 3,1; 12,6] |
13,0 [ 9,3; 20,0] |
0,224 <0,001* 0,687 |
|
|
Баллы эффективности Efficiency |
54,2% |
33,3% |
83,3% |
0,150 0,031* 0,001* |
|
Примечание: n — число обследованных лиц в каждой группе;
p1 — вероятность различий в группах с программами лечения 1 и 2;
p2 — вероятность различий в группах с программами лечения 1 и 3;
p3 — вероятность различий в группах с программами лечения 2 и 3;
* — достоверность различий по критерию Манна-Уитни
для абсолютных величин и по критерию χ² для относительных величин при р<0,05.
Note: n is the number of examined persons in each group;
p1 — probability of differences in groups with treatment programs 1 and 2,
p2 — probability of differences in groups with treatment programs 1 and 3;
p3 — probability of differences in groups with treatment programs 2 and 3;
* — reliability of differences by Mann-Whitney criterion
for absolute values and by χ2 criterion for relative values at p<0.05.
Quantification of the pro-inflammatory cytokine indices, namely IL-1β, IL-6, and TNFα, in the peripheral blood of patients with ACA-HVI revealed significant elevations of their values in all patient groups, attesting to hypercytokinemia. The implementation of three differentiated programs of targeted interferon and immunomodulation therapy had an ambiguous effect on the concentrations of the studied cytokines. In particular, in regard to the pro-inflammatory cytokine IL-1β, which is clearly involved in the immunopathogenesis of herpesvirus infection and the development of neuroimmunoinflammation, its concentration was found to be significantly higher in the blood of patients with ACA-HVI in relation to the control group (p ≤ 0.05) (Fig. 1). After treatment, regardless of the therapy program used, the level of this cytokine decreased significantly in all three groups of patients but never reached the reference values and remained at a significantly higher level.
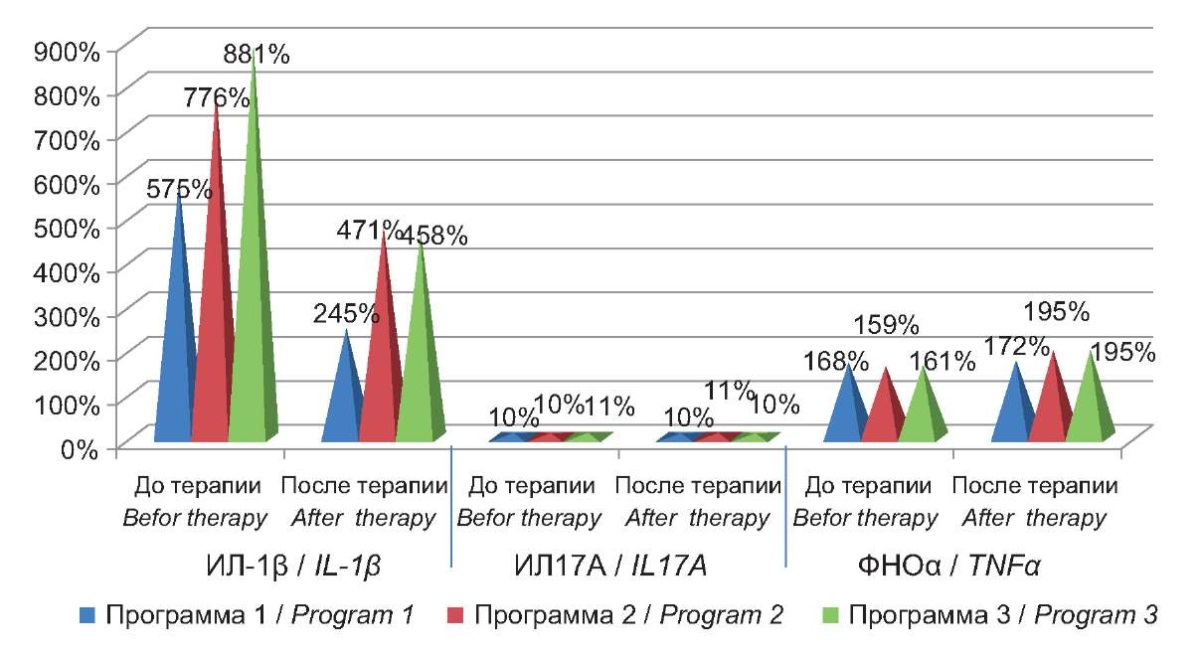
Figure 1. Percentage deviation from control of proinflammatory cytokine levels
in the blood of patients with ACA-HVI before and after therapy.
Рисунок 1. Проценты отклонения от контроля уровней провоспалительных цитокинов
в крови пациентов с АХА-ГВИ до и после терапии.
The concentration of IL-17A produced by T-helper-17 cells in patients with ACA-HVI did not have significant dynamics in response to any of the proposed therapy programs. Quantification of the concentration of pro-inflammatory TNFα revealed a significant increase in its concentration in the blood serum of patients of all three groups before the start of therapy; unfortunately, the integrated program for the correction of the immune system (IPCIS) supplemented by immunomodulatory therapeutic programs proposed by the authors did not significantly affect the dynamics of this indicator.
The decreased levels of concentrations of IFNα and IFNγ in serum before the start of therapy in patients with ACA-HVI significantly increased in response to conducted treatment; more specifically, IFNα increased in response to Programs 1 and 2 while IFNγ elevated as a result of the using all three programs (Fig. 2).
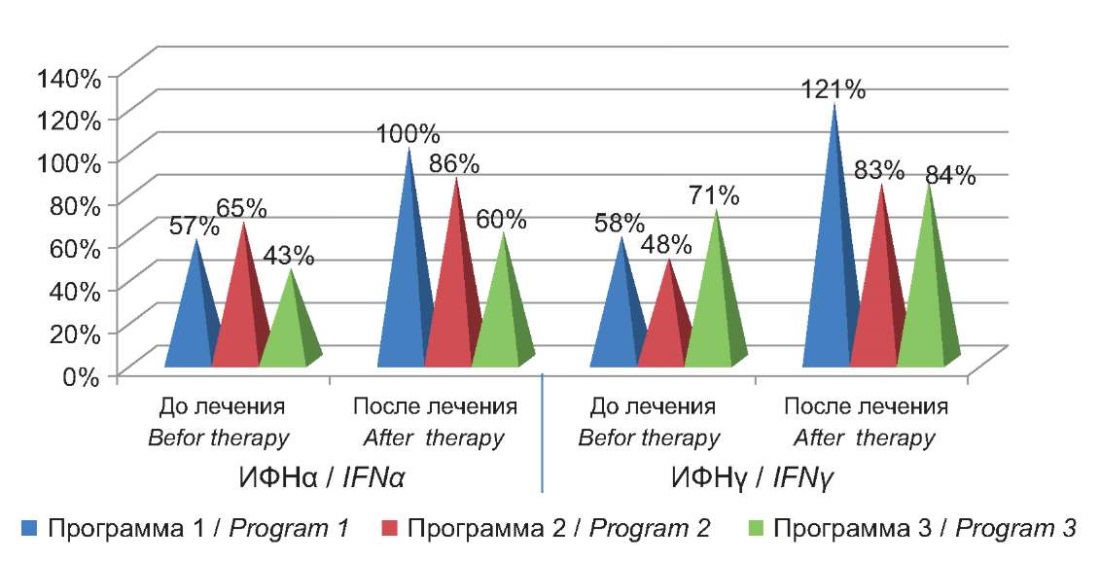
Figure 2. Percentage deviation from the control of interferon levels
in the blood of patients with AСA-HVI before and after therapy.
Рисунок 2. Проценты отклонения от контроля уровней интерферонов
в крови пациентов с АХА-ГВИ до и после проведения терапии.
An assessment of the dynamics on the relative content of NKCs, which were significantly reduced to varying degrees in GI1 and GI2 of patients with ACA-HVI compared to the control group, revealed that therapy according to Program 2 led to a significant increase in their number in comparison with the level before the start of therapy but they still remained below control values (Fig. 3).
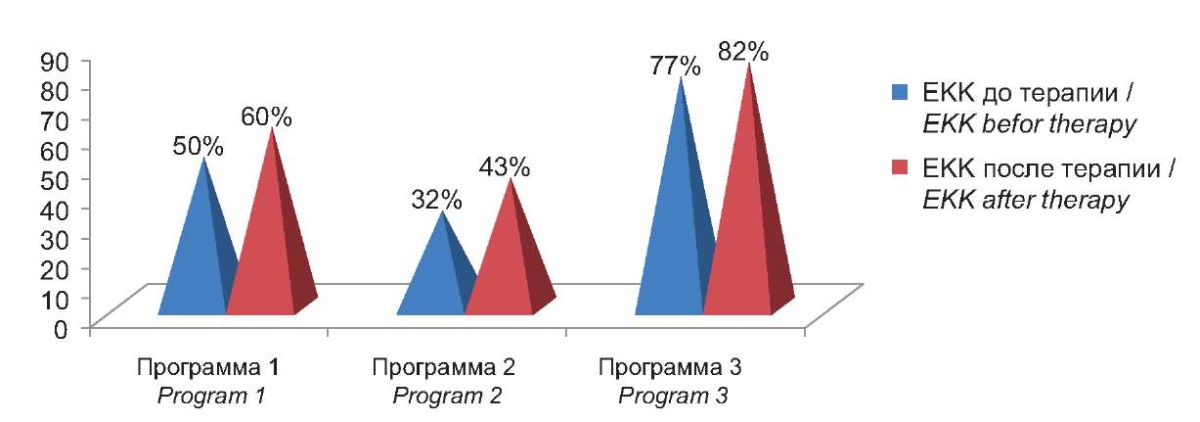
Figure 3. Percentage deviation from control of natural killer cells
in the blood of patients with ACA-HVI before and after therapy.
Рисунок 3. Проценты отклонения от контроля содержания естественных киллерных клеток
в крови пациентов с АХА-ГВИ до и после проведения терапии.
Thus, the implementation of three programs of differentiated targeted interferon- and immunomodulation therapy in accordance with the identified predominant immunophenotype had a true modulating effect on all parts of immunopathogenesis upon ACA-HVI, which was most manifested in the form of the correction of the imbalance in the interferon system trough an increase in the induced production of IFNα and IFNγ and the correction of their concentrations in blood serum, as well as a decrease in the level of pro-inflammatory cytokine IL-1β along with an increase in the number of NKs.
However, the degree of modulating effects varied among different programs as evidenced by a comparison of total scores reflecting the revealed shifts in criterion indices resulting from the implementations of different programs of interferon and immunomodulation therapy. Statistical processing of the data showed that the total score of the efficacy of therapy for Program 1 was 5.0 [ 3.0; 6.0] with the maximum possible value equal to 6 points. Program 2 corresponded to a total score of 3.0 [ 2.0; 6.0]; consequently, its effect was significantly lower than Program 1. The total score of therapy efficacy for Program 3 was 4.0 [ 2.0; 6.0]. The 95% confidence intervals of total scores for assessing the efficacy of the proposed interferon and immunomodulation therapy programs and their ROC analysis are presented in Fig. 4.
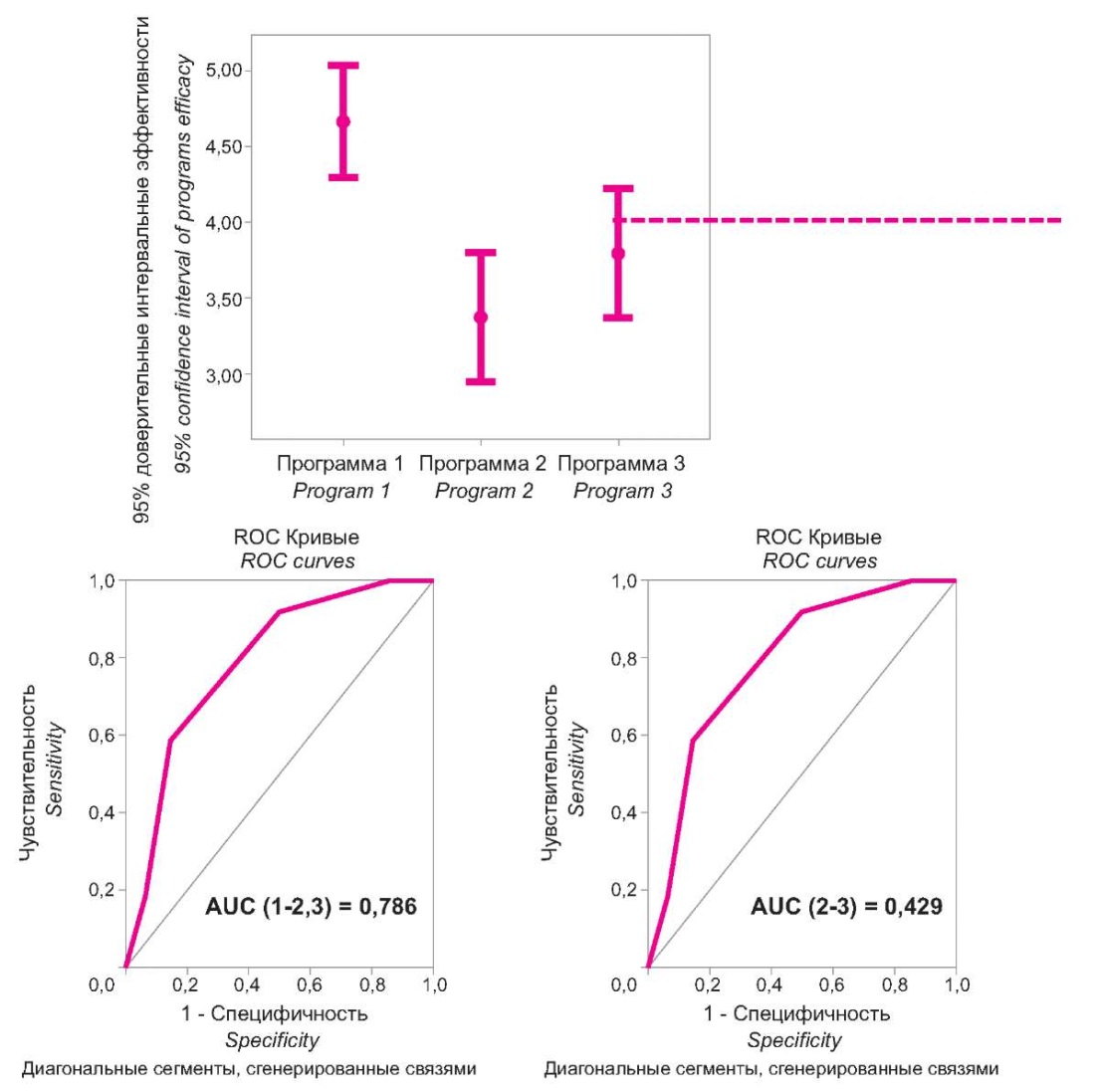
Figure 4. 95% Confidence Intervals of Summary Efficacy Assessment treatment
for each of the proposed programs and ROC curves their diagnostic significance.
Рисунок 4. 95% доверительные интервалы суммарной оценки эффективности лечения
по каждой из предложенных программ и ROC-кривые их диагностической значимости.
The presented graphs clearly demonstrate that the efficacy of Program 1 is the highest with the value of total scores above 4.25. Programs 2 and 3 are rated in the lower total score ranges. The conducted ROC analysis revealed that the significance of the difference in the efficacy of Program 1 was quite high and according to the AUC value (1-2.3), reflecting the comparison of this program with two other programs, amounted to 0.786. The comparison of the efficacies for Programs 2 and 3 identified no significant differences between them; according to the value of AUC = 0.429, the total scores of their assessments did not show any differences.
To assess clinical the efficacy of programs of differentiated interferon and immunomodulation therapy, a comparative analysis of the dynamics of clinical criteria signs of the 1st and 2nd orders was carried out before and after therapy courses (Fig. 5).
Таблица / Table 5
Оценка клинической эффективности программ таргетной интерфероно- и иммуномодулирующей терапии
Evaluation of clinical effectiveness of targeted interferon and immunomodulatory therapy programs
|
Критериальный признак Criteria indicator |
Медиана [минимум; максимум] Median [minimum; maximum] |
|||
|
Программа 1 Program 1 (n = 156) |
Программа 2 Program 2 (n =100) |
Программа 3 Program 3 (n = 79) |
||
|
Частота рецидивов ГВИ (в год) Rate of HVI (per year) |
До лечения Before therapy |
10,0 [ 8,5;14,5] |
10,5 [ 8,0;14,5] |
12,0 [ 10,5;14,5] |
|
После лечения After therapy |
4,5 [ 2,5;6,5] * |
3,5 [ 2,5;5,5] * |
7,5 [ 5,5;9,5] * |
|
|
Длительность рецидива ГВИ (в днях) Duration of HVI (in days) |
До лечения Before therapy |
8,0 [ 6,5;10,5] |
8,0 [ 6,5;10,5] |
9,5 [ 7,5;12,5] |
|
После лечения After therapy |
4,5 [ 3,5; 6,5] * |
5,5 [ 4,5; 6,5] * |
5,5 [ 3,5; 7,5] * |
|
|
Частота ОРВИ (в год) Rate of ARVI (per year) |
До лечения Before therapy |
11,0 [ 9,5; 12,0] |
11,5 [ 10,0; 12,0] |
12,5 [ 11,0;13,5] |
|
После лечения After therapy |
5,5 [ 4,0; 6,5] * |
6,0 [ 4,5;7,0] * |
7,5 [ 6,0;8,0] * |
|
|
Длительность ОРВИ (в днях) Duration of ARVI (in days) |
До лечения Before therapy |
7,5 [ 6,0;8,5] |
7,5 [ 6,0;8,5] |
8,5 [ 7,0;9,5] |
|
После лечения After therapy |
4,5 [ 4,0;5,5] * |
3,5 [ 3,0;4,5] * |
5,5 [ 5,0;7,0] * |
|
|
Длительность периода клин благополучия (в мес.) Duration of well-being period (in months) |
До лечения Before therapy |
6,5 [ 4,5;8,5] |
6,5 [ 4,5;8,5] |
3,0 [ 2,5;5,5] |
|
После лечения After therapy |
10,0 [ 9,5; 12,5] * |
9,5 [ 8,5; 11,5] * |
6,0 [ 5,0; 7,5] * |
|
|
Суммарный балл по Шкале Summary Scale Score |
До лечения Before therapy |
54,5 [ 46,8;62,3] |
46,0 [ 33,0; 55,5] |
52,5 [ 45,0; 50,5] |
|
После лечения After therapy |
30,5 [ 23,0;33,5] * |
21,5 [ 14,0;28,0] * |
31,5 [ 30,5;43,5] * |
|
Примечание: * — достоверность отличий значений до и после проведения терапии.
Note: * — accuracy of differences in values before and after therapy.
Discussion
The data obtained demonstrated the high clinical efficacy of treatment using differentiated targeted interferon and immunomodulation therapy programs in 100% of patients. Patients of GI1 were characterized by the most pronounced positive clinical efficacy, assessed according to clinical criteria of the 1st and 2nd order; among detected indicators, the dominant ones were a 1.8-fold reduction in the duration of relapses of HVI and a 2-fold reduction in the incidence of ARVI related to similar indicators before therapy. Meanwhile, in patients of GI2, the dominant indicators were a decrease in the frequency of relapses of HVI (2.9 times) and in the duration of ARVI (2.1 times) along with the maximum regression of criterion signs of the second order, namely by 2.1 times compared to the indicators before therapy. For the GI3 patients, the dominant clinical effect was related to an increase in the duration of the period of clinical well-being (2.1 times). In all groups of patients, a pronounced regression of clinical signs of chronic fatigue syndrome and cognitive dysfunction were noted; both disorders are based on neuroimmunoinflammation.
Thus, the results of the conducted study demonstrated the high clinical and immunological efficacies for all three programs of differentiated targeted interferon and immunomodulation therapy, oriented towards the identified pathological immunophenotypes and associated clinical manifestations, in immunocompromised patients suffering from ACA-HVI.
Conclusions
The conducted immunological investigations resulted in the identification of variants of pathological immunophenotypes (PIPs), which were characterized by the variability of detected impairments. Specifically, PIP1 was marked by the following features: NG↓ + induced IFNα/IFNγ↓ + CTL↓ + NK↓ + IgM↑ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑); PIP2 was specified by the following characteristics: NG↓+ induced IFNα/IFNγ↓ + NK↓ + IgG↓ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑); and PIP3 involved the following algorithm of alterations: NG↓ + induced IFNα/IFNγ ↓ + hypercytokinemia (IL-1β↑ + IL-6↑ + TNFα↑). The identified impairments in the immune system and the interferon system for each pathological immunophenotype were associated with certain manifestations of the symptoms of ACA-HVI, which were attributable to 3 groups GI1, GI2, and GI3.
The dominant clinical features of GI1 were related to the maximum severity of CFS/ME symptoms and cognitive disorders with 54.5 [ 46.8;62.3] points. For patients of the GI3, the dominant clinical features were specified by the following characteristics: the frequency of HVIs was high with 12.0 [ 10.5;14.5] relapse cases per year and their duration of 9.5 [ 7.5;12.5] days; the frequency of episodes of ARVI was 12.5 [ 11.0;13.5] cases per year with their duration of 8.5 [ 7.0;9.5] days; the period of clinical well-being was minimal with its duration not exceeding 3.0 [ 2.5;5.5] months per year.
For patients of GI1, oriented towards PIP1, treatment based on a combination of 2 strategies turned out to be effective: the first one involved targeted local and systemic interferon therapy using recIFNα2b supplemented with antioxidants, and the second one involved targeted immunomodulatory therapy aimed at restoring CTL and NK deficiencies with a synthetic immunotropic hexapeptide, which is an analog of the active center of the thymus hormone thymopoietin. The clinical efficacy of the therapy was obtained in 100% of patients; immunological efficacy was reached in 89.5% of cases.
For patients of GI2, oriented towards PIP2, the efficacy of the combination of the targeted local and systemic interferon therapy with recIFNα2b supplemented with antioxidants and immunomodulatory therapy aimed at restoring the number of NK cells and correction of neutropenia with glucosaminylmuramyl dipeptide was demonstrated. The clinical efficacy of the therapy was obtained in 100% of patients; immunological efficacy was reached in 57.6% of cases.
For patients of GI3, oriented towards PIP3, the efficacy of targeted local and systemic interferon therapy with recIFNα2b supplemented with antioxidants was shown. The clinical efficacy of the therapy was obtained in 100% of patients; immunological efficacy was reached in 37.5% of cases.
References
1. Baskakova D.V., Khaldin A.A., Birko N.I. Kliniko-epidemiologicheskie kharakteristiki zabolevanij, vyzvannykh gerpesom prostogo gerpesa. Rossijskij zhurnal kozhnykh i venericheskikh boleznej. Prilozhenie «Gerpes». 2006;(2):26–30. (in Russ.).
2. Isakova V.A., Isakov D.V., Arxipova E.I. Gerpesvirusny`e infekcii cheloveka. Rukovodstvo dlya vrachej. St. Peterburg; 2015. (in Russ.).
3. Dyudyun A.D., Polyon N.M., Nagorny O.Ye. Herpesviral infection. The clinical and immunological features. a clinical lecture. Dermatovenerologiya. Kosmetologiya. Seksopatologiya. 2015;3-4:119–125. (in Russ.). eLIBRARY ID: 26697236 EDN: WMFPNR
4. Goreiko T.V., Kalinina N.M., Drygina L.B. The modern conceptions about immunopathogenesis of infection caused by the Epstein–Barr virus. Russian journal of infection and immunity. 2011;1(2):121-130. (in Russ.). eLIBRARY ID: 16382514 EDN: NUMYWX
5. Novikovа I.A., Romanivа O.A. Features of cytokine production in patients with recurrent herpetic infection. Medical Immunology (Russia). 2013;15(6):571-576. (In Russ.) https://doi.org/10.15789/1563-0625-2013-6-571-576
6. Nesterova I.V., Kovaleva S.V., Chudilova G.A., Xalturina E.O., Malinovskaya V.V. Vrozhdyonny`e i priobretyonny`e interferonopatii, associirovanny`e s netipichno protekayushhimi virusny`mi infekciyami i s COVID-19 (monografiya). St. Petersburg: Izdatel`stvo «Dialog»; 2022. (in Russ.).
7. Zhou J, Li J, Ma L, Cao S. Zoster sine herpete: a review. Korean J Pain. 2020;33(3):208-215. https://doi.org/10.3344/kjp.2020.33.3.208
8. Ariza ME. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Human Herpesviruses Are Back! Biomolecules. 2021;11(2):185. https://doi.org/10.3390/biom11020185
9. Shikova E, Reshkova V, Kumanova А, Raleva S, Alexandrova D, et al. Cytomegalovirus, Epstein-Barr virus, and human herpesvirus-6 infections in patients with myalgic еncephalomyelitis/chronic fatigue syndrome. J Med Virol. 2020;92(12):3682-3688. https://doi.org/10.1002/jmv.25744
10. Buckner JH. Translational immunology: Applying fundamental discoveries to human health and autoimmune diseases. Eur J Immunol. 2023;53(12):e2250197. https://doi.org/10.1002/eji.202250197.
11. Khalturina E.O., Nesterova I.V., Markova T.P. An Optimized Program of Targeted Combined Interferono- and Immunotherapy in the Treatment of Atypical Chronic Active Herpesvirus Co-Infections. E`ffektivnaya farmakoterapiya. 2022;18(1):44-49. (in Russ.)
About the Authors
I. V. NesterovaРоссия
Irina V. Nesterova, Dr. Sci. (Med.), Professor, Professor of the Department of Clinical Immunology, Allergology and Adaptology, Medical Institute
Moscow
E. O. Khalturina
Россия
Evgeniya O. Khalturina, Cand. Sci. (Med.), Associated Professor of Microbiology, Virology and Immunology Department named after A. A.Vorobiev
Moscow
Review
For citations:
Nesterova I.V., Khalturina E.O. Efficacy of differentiated targeted interferon and immunomodulatory therapy focused on pathological immunophenotypes in patients with atypical chronic active herpesvirus infections. Medical Herald of the South of Russia. 2024;15(1):165-175. (In Russ.) https://doi.org/10.21886/2219-8075-2024-15-1-165-175
JATS XML









































